Container closure integrity evaluation of molded vials stored at -80°C during using laser-based headspace analysis
Increasing flexibility in pharmaceutical packaging for aseptic filling
Navigating the regulatory landscape for cannabidiol (CBD) packaging
Kindersichere Verpackungen
Glass Packaging Case Study - Rethinking the pharma packaging paradigm: A case for molded glass
Sprache :English Download - (0.97 Mo)Press release
SGD Pharma expands North American pharma glass offering with Prince Sterilization Services, LLC, debuting My Sterinity Nasal at DCAT Week
Sprache :English Download - (0 Mo)SGD Pharma showcases Sustainable Glass Innovation and Decorative Excellence at Paris Packaging Week – PCD 2026
Sprache :English Download - (0 Mo)SGD Pharma leads pharma packaging innovation and decarbonization at Pharmapack Europe 2026
Sprache :English Download - (0 Mo)SGD Pharma Zhanjiang Becomes China’s First Glass Manufacturer to Achieve ISO 14021 Certification for PCR Packaging
Sprache :English Download - (0 Mo)SGD Pharma presents new innovations at CPHI Frankfurt 2025
Sprache :English Download - (0 Mo)Corning and SGD Pharma Inaugurate Glass Tubing Facility in Telangana To Expand Pharmaceutical Packaging Manufacturing
Sprache :English Download - (0 Mo)A Strategic Step Forward: Alphial S.r.l. Joins SGD Pharma
Sprache :English Download - (0 Mo)SGD Pharma releases new sustainability report outlining industry-leading pharmaceutical glass CSR performance
Sprache :English Download - (0 Mo)SGD Pharma showcases its SEALIAN platform at Pharmapack Europe 2025
Sprache :English Download - (0 Mo)SGD Pharma spotlights new optimized solutions at CPHI worldwide 2024, continuing its leadership in sustainability and innovation
Sprache :English Download - (0 Mo)SGD Pharma showcases new post-consumer recycled glass packaging at Luxe Pack Monaco 2024
Sprache :English Download - (0 Mo)SGD Pharma held its European Suppliers Day bringing together about 100 stakeholders of the glass and sustainability ecosystem on June 18th
Sprache :English Download - (0 Mo)SGD Pharma releases its latest sustainability report underscoring its global commitment to the highest environmental, social and governance standards
Sprache :English Download - (0 Mo)SGD Pharma invests €20M to build furnace and upgrade industrial facility at its Zhanjiang plant in China, advancing its decarbonisation roadmap
Sprache :English Download - (0 Mo)SGD Pharma announces the appointment of Philippe Pourquery as Group Financial Officer and member of the Executive Committee
Sprache :English Download - (0 Mo)SGD Pharma successfully trials hydrogen burners at its Saint-Quentin-Lamotte (SQLM) plant to accelerate its ambitious decarbonization roadmap
Sprache :English Download - (0 Mo)SGD Pharma awarded 2024 gold EcoVadis rating in recognition of its commitment to sustainability
Sprache :English Download - (0 Mo)SGD Pharma optimizes global fill and finish operations with the introduction of its new range of Type I tubular vials featuring Corning’s Velocity® Vial coating technology for enhanced product quality and patient safety
Sprache :English Download - (0 Mo)SGD Pharma expands capacity of siliconized glass vials with the announcement of a new siliconization line at its Center of Excellence for Type I glass in France
Sprache :English Download - (0 Mo)SGD Pharma announces the appointment of Fabio Invernizzi, General Manager West Business Unit
Sprache :English Download - (0 Mo)SGD Pharma defines 2024 growth strategy as it strives for market leadership with investment achievements
Sprache :English Download - (0 Mo)EUROPEAN CONTAINER GLASS FEDERATION NEW LEADERSHIP TEAM APPOINTED AT FLACONNAGE BOARD OF DIRECTORS
Sprache :English Download - (0 Mo)SGD Pharma unveils NOVA lightweight glass bottle packaging innovation, addressing the commitment to environmental responsibility from cosmetic and beauty brands Paris
Sprache :English Download - (0 Mo)SGD Pharma commits to near-term Science Based Targets initiative as part of sustainability drive
Sprache :English Download - (0 Mo)SGD Pharma to present its high-end glass packaging innovations tailored to the cosmetics and beauty industry at Luxe Pack Monaco 2023
Sprache :English Download - (0 Mo)SGD Pharma’s latest annual sustainability report details global environmental, social and ethical efforts
Sprache :English Download - (0 Mo)Corning and SGD Pharma announce joint venture to open new glass tubing facility and expand access to Corning® Velocity® Vial technology in India
Sprache :English Download - (0 Mo)Continued focus on innovation to be presented by SGD Pharma at Pharmapack Europe 2023
Sprache :English Download - (0.15 Mo)Committed to staying ahead with pioneering glass packaging solutions that answer the changing needs of the pharmaceutical industry, SGD Pharma is set to showcase its latest innovations at CPhI worldwide 2022
Sprache :English Download - (0 Mo)SGD Pharma returns to Luxe Pack Monaco with new proposition for premium glass packaging for cosmetics and beauty
Sprache :English Download - (0.14 Mo)SGD Pharma announces new CEO as it targets further growth
Select your language:SGD Pharma expands cosmetics glass packaging offering, appoints industry leader
Sprache :English Download - (0.15 Mo)SGD Pharma launches industry first Ready-to-Use sterile 100 ml molded glass vials in SG® EZ-fill® packaging technology
Select your language:2022 Année Internationale du Verre, une double cérémonie d’ouverture à l’ONU à Genève et au Palais du Luxembourg à Paris
Sprache : Download - ( Mo)SGD Pharma speeds up time to market with industry first Ready-to-Use SG EZ-fill® Nest & Tub packaging solution for molded glass vials for parenteral drugs
Select your language:Key milestone in molded glass: SGD Pharma elevates Type I offering with three tailored product and service solutions
Select your language:SGD Pharma takes lead in sustainability, awarded Platinum EcoVadis rating for the first time
Select your language:SGD Pharma continues customer-led innovation with range extension for Sterinity Ready-to-Use molded glass vials: adds amber 50ml size ISO and EasyLyo in Tray
Select your language:Q&A
- All topics
- COVID-19
- Allgemeines
- Herstellung von Flaschen
- Investition & Zukunftssicherheit
- Kontinuierliche Verbesserungen an geformtem Typ 1 Glas
Operation Marketing & Communication Manager |
Alice ACHACHE
Media partner | The Scott Partnership
Ja. Unsere hochmoderne Produktionsanlage in Vemula bei Hyderabad in Indien, die 2013 fertiggestellt wurde, ist für die Herstellung von Typ-I-Glas registriert, das mit den Pharmakopöen von Japan, Europa, Indien und den USA konform ist. Die in Vemula hergestellten Klassenbesten Röhrenflaschen und Ampullen werden in Indien für die Verpackung von COVID-19-Impfstoffflaschen verwendet. Neben der lokalen und nationalen Versorgung mit Röhrenflaschen verfügt der Standort Vemula auch über einen großen Ausstoß an geformtem Typ-I-Glas für die Märkte in Asien, EMEA und den USA und wird als Teil der globalen Lieferkette für viele pharmazeutische Unternehmen eingesetzt.
SGD Pharma ist ein weltweit führender Hersteller von pharmazeutischen Glasverpackungen, der die Anforderungen einer Vielzahl von Märkten erfüllt, darunter parenterale, nasale und orale Medikamente sowie für Kosmetik und Nahrungsmittel.
Zwei Werke sind Best-in-Class-Anlagen, die sich auf Typ-I-Glas spezialisiert haben (Saint- Quentin-Lamotte (SQLM) in Nordfrankreich und Vemula in Indien). SQLM ist ein Center of Excellence für Typ-I-Glas. Typ-II-Glas für Anästhetika wird in Sucy-en-Brie, Frankreich, und Kipfenberg, Deutschland, hergestellt. Typ III-Braunglas für Analgetika wird in Sucy-en-Brie hergestellt. Alle Produktionsstätten sind nach ISO 15378 zertifiziert. Vemula, Indien, ist außerdem nach ISO 14001 und 18001 akkreditiert.
Unser Ziel ist es, die Gesundheit von Patienten zu verbessern und zu schützen, indem wir unseren pharmazeutischen Kunden qualitativ hochwertige, zuverlässige und innovative Primärglasverpackungen anbieten.
Zwischen 2020 und 2025 werden wir alle unsere 7 Öfen umbauen und mit neuen Technologien aufrüsten (Live-Überwachungssysteme, bessere Wärmedämmung, erhöhte elektrische Verstärkung). Diese Verbesserungen werden uns schließlich in die Lage versetzen, unsere Gesamtenergieeffizienz bis 2025 um mindestens 7 % zu verbessern.
Im Jahr 2019 haben wir ein großes Werksentwicklungsprojekt in Sucy-en-Brie, Frankreich, begonnen. Diese Investition in Höhe von insgesamt 31 Mio. € ist die größte Investition von SGD Pharma seit dem Bau des Werks in Saint-Quentin-Lamotte. Das Werk in Sucy-en-Brie, das bereits nach ISO 15378 & 50001 zertifiziert ist und über 8 Produktionslinien verfügt, die mit ISO 8 Reinräumen verbunden sind, wird zu einer neuen Generation von Pharma- Glasverpackungseinheiten gehören.
Das Projekt umfasst den kompletten Umbau und die Aufrüstung des Ofens 2, den Umbau des Gebäudes selbst, die vollständige Überarbeitung der ISO 8-Reinräume, IS-Maschinen der neuen Generation und die Installation der modernsten Technologien für die automatische Produktinspektion. Dank Live-Überwachungssystemen, besserer Wärmedämmung und verstärkter elektrischer Verstärkung werden die CO2-Emissionen um 10 % reduziert. Die Neugestaltung der Verkehrsflächen wird zudem die Sicherheit der Anlage verbessern.
Unsere Typ I Glasflaschen sind widerstandsfähig und chemisch inert (dank ihrer hohen hydrolytischen Beständigkeit). Damit sind sie eine relevante Lösung, um das Angebot an Röhrenglasflaschen des Typs I zu ergänzen und auf diesen beispiellosen Nachfrageschub zu reagieren. Sie sind ideal sowohl für die Lagerung als auch für den Transport von Impfstoffen auf der ganzen Welt.
SGD Pharma hat sich zum Ziel gesetzt, seine CO2-Emissionen um 5 % pro Tonne produzierter Ware zu reduzieren. Dies entspricht einer Einsparung von über 10.000 Tonnen CO2 bis 2025. Wir planen dies zu erreichen über:
- Teilnahme an der FEVE-Initiative "Ofen der Zukunft": Wir sind eines von 20 Unternehmen aus der Glasindustrie, die sich zusammengeschlossen haben, um ein Pilotprojekt für einen Schmelzofen zu entwickeln, zu finanzieren und zu testen. Dies wird der weltweit erste groß angelegte Hybrid-Oxy-Brennstoff-Ofen sein, der mit 80 % erneuerbarem Strom betrieben wird. Dies wird die derzeitigen fossilen Energiequellen ersetzen und die CO2-Emissionen um 50 % reduzieren.
- Investition in die Umweltleistung: Um unser Energie-Engagement zu demonstrieren, haben wir 2019 drei Werke nach ISO 14001 (Umweltmanagement) zertifizieren lassen (Kipfenberg, Zhanjiang und Vemula) und planen, die verbleibenden Werke (Sucy-en-Brie und Saint-Quentin) bis Ende 2021 zu zertifizieren.
- Investition in den Wasserverbrauch: Um unseren Wasserverbrauch zu reduzieren, haben wir an allen unseren Produktionsstandorten geschlossene Kreisläufe installiert.
Im Jahr 2019 haben wir unseren Gesamtwasserverbrauch im Vergleich zu 2018 um 18 % reduziert. Dadurch konnten wir mehr als 156 000 m3 Wasser einsparen, was hauptsächlich durch die hervorragende Leistung unseres französischen Werks in Sucy-en-Brie erreicht wurde.
- Investition in die Wiederverwendung von Glas: Close the Glass Loop ist eine europäische Bottom-up, kollaborative, öffentlich-private Partnerschaft, die darauf abzielt, die Quantität und Qualität des verfügbaren recycelten Glases zu erhöhen. Diese Initiative folgt dem europäischen Green-Deal-Rahmenwerk, das kürzlich die Ziele für die Recyclingquote geändert hat, um den Übergang zu einer stärker kreislauforientierten Wirtschaft zu beschleunigen.
Es wurden zwei Hauptziele festgelegt:
- Anhebung der Glassammelquote auf 90 % bis 2030 (von 76 % im Jahr 2019)
- Verbesserung der Qualität von recyceltem Glas, damit mehr recycelte Inhalte in einem neuen Produktionskreislauf verwendet werden können, besser, gemeinsam.
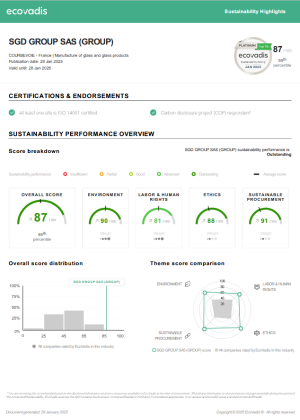
Die Bemühungen von SGD Pharma im Bereich Corporate Social Responsibility (CSR) wurden von EcoVadis, einem unabhängigen Gutachter, anerkannt, der uns im Jahr 2020 ein Platin-Rating verliehen hat - damit gehört SGD Pharma zu den besten 1% aller bewerteten Unternehmen. Diese Bewertung zeigt das Engagement von SGD Pharma für nachhaltige Geschäftspraktiken und unsere Ausrichtung auf globale Maßnahmen zum Schutz der Umwelt und zur Bewältigung sozialer Probleme. Außerdem erhielten wir vom CDP eine B- Score-Karte als Anerkennung für unsere Maßnahmen zur Bekämpfung des Klimawandels.
Zu den Kunden von SGD Pharma gehören globale Pharma-, Biopharma-, Generika- und Impfstoffhersteller sowie die Kosmetikindustrie (insbesondere in China).
Im Jahr 2020 hat SGD Pharma den täglichen Betrieb fortgesetzt und sogar die Leistung in allen Werken verbessert, das Vertrauen der Kunden gestärkt und Marktanteile gewonnen, die F&E-Kapazitäten beschleunigt und Mitarbeiter eingestellt und geschult. Unsere Werke in Asien und Europa unterliegen alle den höchsten Sicherheitsmaßnahmen, um die Gesundheit und Sicherheit der Mitarbeiter zu gewährleisten, während der Produktionsbetrieb weiterläuft.
Die Herstellung und der weltweite Versand von Impfstoffen erfordern eine gut geführte Logistik und den Zugang zu einer kontinuierlichen und gesicherten Versorgung mit Hilfsstoffen, um sicherzustellen, dass es bei der Impfstoffverteilung keine Engpässe gibt. Als Unternehmen für Primärverpackungen aus Glas arbeiten wir unermüdlich daran, diesen Zugang für die weltweiten COVID-19-Immunisierungskampagnen zu gewährleisten.
Wir produzieren über 8 Millionen Flaschen pro Tag, verteilt auf Werke in Europa und Asien. Pro Jahr werden mehr als 2 Milliarden Flaschen hergestellt und verkauft.
Um diesen Impfstoff für den weltweiten Vertrieb vorzubereiten, bietet SGD Pharma den Zugang zu unserer gesamten Produktion in unserem hochmodernen, nach ISO 15378 zertifizierten Werk in der Normandie, Frankreich, an, wo unsere tägliche Kapazität bei 400 bis 500Ku von international anerkannten, neutral geformten Glasflaschen vom Typ I liegt, die in vielen Größen erhältlich sind, darunter 10ml, 20ml, 30ml und 50ml.
Seit 1896: Wir schöpfen aus dem mehr als 100-jährigen Know-how, das wir von Saint-Gobain erlangt haben, und sind weltweit als technischer Marktführer für Glasverpackungen bekannt.
Wir bieten eine breite Palette an Größen und Veredelungen in Typ I, II oder III, Braunglas und Klarglas sowie innovative Mehrwertdienste wie Innensilikonisierung oder Kunststoffschutzbeschichtungen.
Wir bauen langfristige Beziehungen zu unseren Kunden auf und sind reaktionsschnell, zuverlässig, transparent und äußerst kooperativ. Wir arbeiten eng mit unseren Kunden zusammen und erforschen die neuesten Technologien. Wir suchen nach spezifischen Herausforderungen, die wir durch die Erforschung oder Anwendung neuer Technologielösungen angehen.
Wir beschäftigen über 3.000 Experten in den Bereichen F&E, Vertrieb, Marketing, Qualität, Regulierung, Umwelt, Gesundheit und Sicherheit und Produktion. Unsere Teams arbeiten zusammen, um schon heute die Herausforderungen von morgen zu lösen.
5 Produktionsstätten in 4 Ländern (Frankreich, Indien, Deutschland, China). Wir kombinieren die Kontrolle des Glasprozesses mit der Umsetzung von GMPs für pharmazeutische Primärverpackungen in allen unseren Produktionsstätten.
Nach der Fertigstellung unseres Werks in Vemula im Jahr 2013 und von SQLM im Jahr 2016 planen wir, weiterhin in die neueste Technologie für unsere Anlagen zu investieren und unseren Kunden eine unerschütterliche Produktqualität zu bieten. Dies zeigt sich in unseren kommenden Produktinitiativen für das Jahr 2021, zu denen auch die Aufwertung unseres Produktportfolios für geformtes Typ-I-Glas gehört, um unseren Kunden Lösungen zu bieten, die ihre individuellen Geschäfts- und Produktanforderungen erfüllen. Durch kontinuierliche Verbesserungen der Glaszusammensetzung, der Produktionsprozesse und -anlagen sowie der Qualitätskriterien bleibt SGD Pharma führend im Bereich der Behälterglasverpackungen.
Unser SQLM-Standort in der Normandie, Frankreich, verfügt über eine neue Betriebsstätte, die 2013 eröffnet wurde, und die hochmoderne Anlage von Vemula in Indien wurde im Februar 2018 fertiggestellt, mit neuen Öfen, jeweils mit ISO-8-Reinräumen (1700 m2 und 2400 m2) Zusammen bieten diese Anlagen 31.800 m2 und 11.000 m2, was eine Produktionskapazität von 1,5 Millionen Fläschchen und Ampullen pro Tag von SQLM und 1 Million Fläschchen pro Tag von Vemula ergibt. Zusammen mit unseren Lagerkapazitäten ermöglicht uns dies, die Nachfrage nach Impfstoffen zu decken und gleichzeitig unsere anderen Kunden zu bedienen.
Borosilikatglas Typ I wird für die Verpackung von parenteralen Medikamenten verwendet. Es ist das ideale Material aufgrund seiner Inertheit und chemischen Stabilität, die Wechselwirkungen mit dem Medikament begrenzt und dessen Wert sowie die Patientensicherheit schützt.